 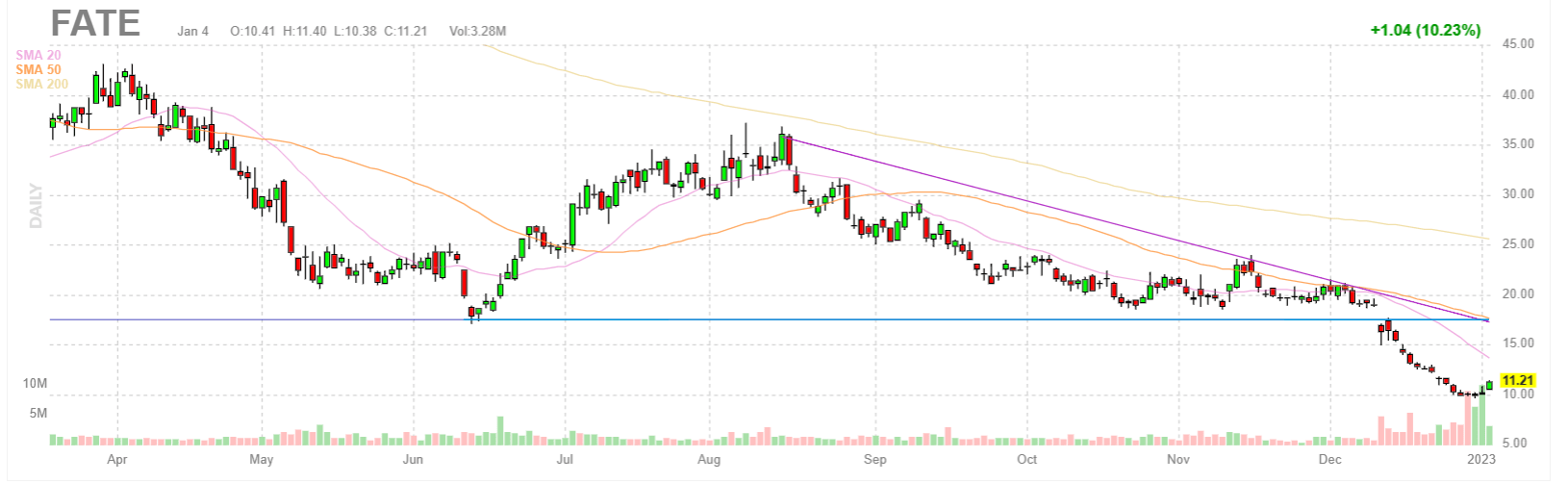
EtO processing is a gas sterilization process in which pallets of packaged goods are loaded into a chamber that is then injected with EtO gas to penetrate the packaging. The Company’s sterilization services rely on three primary technologies, one of which is Ethylene Oxide (“EtO”) processing. Terminal sterilization is the process of sterilizing a product in its final packaging. Through its Sterigenics brand, which accounts for the majority of Sotera’s annual revenues, Sotera provides outsourced terminal sterilization services for the medical device and pharmaceutical markets. The Company operates through three businesses: Sterigenics, Nordion, and Nelson Labs. Sotera provides sterilization and lab testing and advisory services to the medical device and pharmaceutical industries. On this news, Fate’s stock price fell $6.76 per share, or 61.45%, to close at $4.24 per share on January 6, 2023.Īs a result of Defendants’ wrongful acts and omissions, and the precipitous decline in the market value of the Company’s securities, Plaintiff and other Class members have suffered significant losses and damages.įor more information on the FATE class action go to: Ĭlass Period: Pursuant to the NovemIPO Pursuant to the MaSPO and/or NovemSeptember 19, 2022 On January 5, 2023, after the markets closed, Fate issued a press release announcing that it had terminated the Janssen Collaboration Agreement. Specifically, the Company disclosed that it was “not able to align with Janssen on their proposal for continuation of our collaboration, where two product candidates targeting high-value, clinically-validated hematology antigens were set to enter clinical development in 2023” As a result of the termination, Fate revealed that all licenses and other rights granted pursuant to the Janssen Collaboration Agreement would terminate, that it would reduce its headcount to about 220 employees in Q1 2023, and that it would discontinue several of its natural cell killer programs in various cancers, including FT516 and FT538 NK cell programs in acute myeloid leukemia, FT516 and FT596 NK cell programs in B-cell lymphoma, and FT538 and FT536 NK cell programs in solid tumors. Specifically, Defendants made false and/or misleading statements and/or failed to disclose that: (i) the Janssen Collaboration Agreement was less sustainable than Fate had represented to investors (ii) accordingly, certain the clinical programs, milestone payments, and royalty payments associated with the Janssen Collaboration Agreement could not be relied upon as future revenue sources (iii) as a result, Fate had overstated the impact of the Janssen Collaboration Agreement’s on Fate’s long-term clinical and commercial profitability and (iv) as a result, the Company’s public statements were materially false and misleading at all relevant times. 
Throughout the Class Period, Defendants made materially false and misleading statements regarding the Company’s business, operations, and compliance policies. On the news, Fate’s stock price jumped 8.8% in trading on April 3, 2020. In addition, Fate was eligible for up to $3 billion in various milestone payments and double-digit royalties on any net sales from the collaboration. (“Janssen”), one of the Janssen Pharmaceutical Companies of Johnson & Johnson, for cell-based cancer immunotherapies, under which Fate received a $50 million upfront payment (the “Janssen Collaboration Agreement”). On April 2, 2020, after the market closed, Fate announced its entry into a global collaboration and option agreement with Janssen Biotech, Inc.  
Fate is a clinical-stage biopharmaceutical company that develops programmed cellular immunotherapies to treat cancer and immune disorders. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
